Cell therapy Solutions
ⅰ.
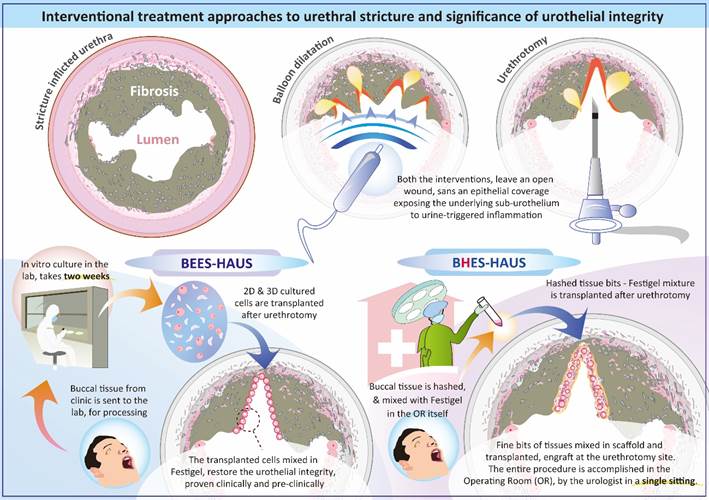
The BEES-HAUS (Buccal Epithelium Expanded and Encapsulated in Scaffold–Hybrid Approach) procedure for urethral stricture has been successfully tech- transferred to Japan and implemented under the Act on the Safety of Regenerative Medicine of Japan following extensive preclinical validation in Japan demonstrating engraftment morphologically and immunohistochemically as well as a pilot clinical study with documented safety and long-term outcome.
(Image source: Front. Bioeng. Biotechnol., 27 November 2025
Volume 13 – 2025 | https://doi.org/10.3389/fbioe.2025.1687741)
ⅱ.
A simplified BHES-HAUS (Buccal Epithelium Hashed and Encapsulated in Scaffold – Hybrid Approach to Urethral Stricture)approach has now been developed and the preliminary results of its clinical trial has demonstrated safety and promising outcome. This work has been accepted for presentation at American Urology Association (AUA) 2026 Annual Meeting.
ⅲ.
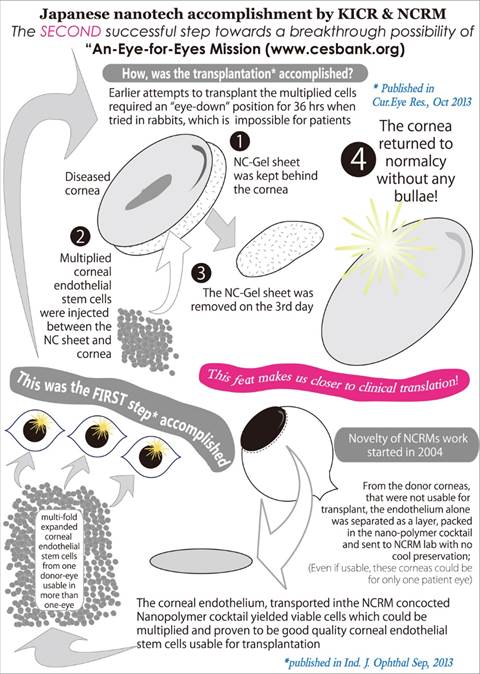
For corneal endothelial diseases such as bullous keratopathy, a novel approach called ICE-TUBE (In vitro Expanded Corneal Endothelial Precursors Transplanted Using BLIS-Film: Eye-for-Eyes Mission)has been developed. This technique enables corneal endothelial precursor cells isolated from a single cadaver donor eye to be expanded in vitro and transplanted, using a nanocomposite gel scaffold (BLIS-FILM: Bio-inert Low IgG Sensitive Film), into more than one recipient eye. This “Eye-for-Eyes” mission proven successfully in vitro and in a pilot clinical study with 16-year follow-up aims to address the global shortage of donor corneas.
ⅳ.
For unilateral limbal stem cell deficiency or ocular surface damage affecting one eye, an innovative method of ocular surface reconstruction called LEES-HARC (Limbal Epithelium Expanded and Encapsulated in Scaffold–Hybrid Approach to Regenerate Cornea) has been developed. This technique demonstrated in a pre-clinical animal model involves the expansion of limbal stem cells from the unaffected healthy eye using Festigel (Free-from-Endotoxin Scaffold of Thermoresponsive Intelli-GEL), which serves as a supportive scaffold during transplantation.
ⅴ.
EELS-TALC (Enriched with Essentials and Lapped in Scaffold Transplant-suitable Autologous Leveraged Chondrocytes)is a novel technology that enables the expansion of autologous chondrocytes, even from elderly individuals, and their transplantation using a Festigel scaffold, while maintaining the hyaline cartilage phenotype, as demonstrated in in vivo animal models. Cells tissue-engineered in vitro using EELS-TALC have been shown to enhance resident chondroprogenitors and mesenchymal stem cells, help retain hyaluronic acid (HA), increase microRNA-140 (miRNA-140) expressionand exhibit reduced expression of senescence-associated markers, resulting in a beneficial reversal of cellular aging without any genomic modifications.

ⅵ.
Autologous Immune Enhancement Therapy (AIET)for cancer is a novel autologous cell-based immunotherapy approach in which a patient’s own peripheral blood is collected and natural killer (NK) cells and T lymphocytesare expanded & activated in the laboratory for 16–21 days which are then reinfused into the patient to target and destroy cancer cells. AIET has been in clinical practice in Japan for more than two decades with randomized clinical trialsestablishing safety and has been successfully transferred by GN Corporation to Malaysia and Vietnam. This least toxic therapeutic modality for cancer is applied as an approved procedure under Japan’s Act on the Safety of Regenerative Medicineand when combined with conventional treatment approaches for cancer such as chemotherapy, radiotherapy & surgery, enhances the efficacy by 20-30%.




